Facts & Figures
Scientific Services
Support for researchers with ideas for better patient care.
Consulting
Number of contact requests
493
contact requests were sent to DKF Scientific Services.
This represents a decrease of 4 per cent compared to last year.
Around 31 per cent of the requests could be answered directly in writing or verbally. For all others, personal consultations were arranged.
Number of consulting sessions according to services

541
consulting sessions in total.
This represents a decrease of 28 per cent compared to last year, which is mostly due to a shift of resources in favour of project support activities.
General project consulting (initial contact)
General project consulting (follow-up)
Patient & Public Involvement
Quality Management
Data Science consulting hour
Data Management consulting hour
Statistics consulting hour
Project Support
Number of clinical studies and research projects according to HRA classification

287
clinical studies and research projects in total.
This represents an increase of 7 per cent compared to last year.
60 per cent were ongoing projects started before 2025, 40 per cent of projects were newly started in 2025.
Clinical trials with medicinal products
Clinical trials with transplant products
Clinical trials with medical devices
Other clinical trials
Human research projects involving further use of biological material and health-related personal data
Human research projects involving sampling of biological material or collection of health-related personal data
Research projects without classification according to the Human Research Act (HRA)
other
HRA - Human Research Act, Federal Act on Research involving Human Beings
Percentage of involvement of scientific services supporting 287 clinical studies and research projects

Data Analysis/Statistics
Data Management
Data Science
Project Management
Monitoring
On Site & Safety Management
Regulatory Affairs
IT Services
Quality Management
Communication
31'700
working hours spent in total.
This represents an increase of 3 per cent compared to last year.
Clinical Databases
Number of active databases according to software

261
active databases in total.
This represents an increase of 32 per cent compared to last year, which is primarily due to an increasing demand for straightforward REDCap® databases.
secuTrial® databases newly set up in 2025
secuTrial® databases set up before 2025
REDCap® databases newly set up in 2025
REDCap® databases set up before 2025
Number of participants newly administered according to software

in secuTrial® databases
in REDCap® databases
40'814
new participants in total.
This represents an increase of 9 per cent compared to last year.
Registrations & Submissions
Number of studies* registered on clinicaltrials.gov since 2018

Registered by DKF Scientific Services
Registered by research groups directly and administered by DKF Scientific Services
64
new registrations in total. Since 2018, a total of 582 studies have been registered.
* This graph considers studies conducted at the University Hospital Basel only. DKF Scientific Services is mandated by the University Hospital Basel management to register and administer clinical studies in clinicaltrials.gov since 2018.
Number of submissions to ethics committees and competent authorities

143
submissions in total.
This represents a decrease of 27 per cent compared to last year, which is mainly due to a higher number of complex submissions and a trend towards researchers submitting simple studies by themselves without expert support.
Submissions to ethics committees and competent authorities in Switzerland
Submissions to international ethics committees and competent authorities
Monitoring
& Safety
Management
Number of monitored studies according to number of centres and category
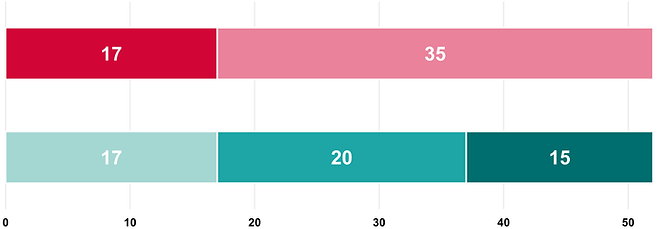
Monocentric studies
Multicentric studies
ClinO/ClinO-MD category A studies
ClinO/ClinO-MD category B studies
ClinO/ClinO-MD category C studies
52
studies in total.
This represents an increase of 2 studies compared to last year.
ClinO... Ordinance on Clinical Trials with the exception of Clinical Trials of Medical Devices
ClinO-MD... Ordinance on Clinical Trials of Medical Devices
Number of monitoring visits at national and international study centres

182
visits in total.
This represents a decrease of 9 per cent compared to last year.
International study centres
Study centres in Switzerland
Number of studies with Medical Monitoring and/or MedDRA Coding services
14
studies in total.
This represents an increase of 4 additional studies compared to last year.
Project
Management
Number of studies supported with project management services

Monocentric studies
Multicentric studies
National studies
International studies
44
studies in total.
This represents an increase of 16 per cent compared to last year.
Number of study centres supported with project management services

493
study centres in total.
This represents an increase of 9 per cent compared to last year.
Study centres in Switzerland
Study centres in Europe
Study centres in the rest of the world
Quality Affairs
Number of registered users in Orca*

371
new users registered in total.
Overall, 1'884 users have been registered over the past years.
Users from DKF research groups
Users from external institutions**
Users from DKF Scientific Services, ASZ, Training & Education and staff
*Orchestrate your Clinical Research Activities (Orca) is a web-based quality management tool for academic clinical research developed and operated by DKF Scientific Services
**University Hospital Zurich, University Children’s Hospital Zurich, University Psychiatric Hospital Zurich, Zurich City Hospital, Insel Gruppe Berne, Department of Clinical Research Berne, University Clinic Balgrist, Schulthess Clinic, Medical Image Analysis Center Basel, Swiss Tropical and Public Health Institute, Cantonal Hospital St. Gallen, Research Cluster Molecular and Cognitive Neurosciences University of Basel, Institute of Molecular and Clinical Ophthalmology (IOB) Basel, University Geriatric Medicine Felix Platter Basel, University Children’s Hospital Basel, Reconnect Labs AG, Reha Rheinfelden, Zenon Therapeutics AG,CTU Swiss Paraplegic Centre Nottwil
Support before, during and after inspections and audits

10
inspections and audits in total.
This represents one inspection less and two audits more compared to last year.
Inspections by Swissmedic
Audits by the Ethics Committee of North-Western and Central Switzerland
Audit by the Federal Office of Public Health FOPH
Audit by a sponsor
